The overlap probability and extent, which determines the overall stabilization, falls off very quickly with the separation between the overlapping orbitals - usually beyond one bond it is very weak, and beyond 3 it is negligible. "Delocalization" quite literally means that the $\pi$-electrons, instead of being associated with a particular bond between 2 atoms of the molecule, are part of an extended $\pi$-cloud over the molecule (an extended molecular orbital formed by the overlap of partially- and fully-occupied molecular orbitals and unoccupied atomic orbitals (near the $\sigma$-bonds) - the premise of Molecular Orbital Theory - please do find the time to read about it!). 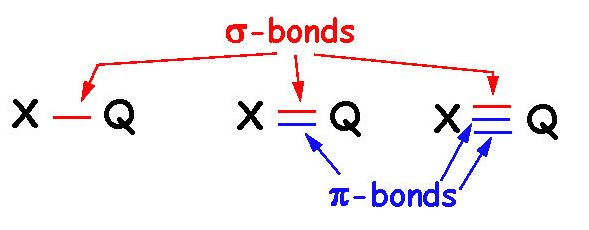
The rearrangement is to a conjugated diene because conjugation facilitates resonance - if the $\pi$-bonds are too far apart, the $\pi$-clouds cannot overlap, which is resonance (or electron delocalization over $\pi$-bonds). This "smoothed-out" compound usually has more symmetry - and in most cases, symmetrical configurations are more stable than asymmetrical ones if both are under comparable amounts of steric stress. Resonance-stabilized compounds have lower energy than non-resonance stabilized ones, but why is that? One of the possible explanations is that resonance "averages out" energy minima and maxima between the resonating parts of the compound. It is indeed due to the resonance-stabilization of the conjugated products formed due to the rearrangements you quote. I found points to be correct, but the explanation to be quite less than ideal, so to speak, so I'll try to provide an alternative and I hope somewhat more sensible explanation as to why this happens. Has already proposed a possible mechanism, and has tried to explain why it might happen.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
